The next dimension in drug discovery
VAST TECHNOLOGY
VAST (Versatile Assembly on Stable Templates) is a chemistry-based technology that provides a turnkey solution to access drug-like molecules on a diverse set of biologically-relevant 3D scaffolds.
VAST uses one of nature’s 3D building blocks, the carbohydrate pyranose, to build a library of drug-like molecules with with functionality attached to five different chiral centres on the pyranose ring. This enables the synthesis of highly diverse 3D structures as opposed to the 2D molecules typically found in the small-molecule compound collections of pharmaceutical companies.
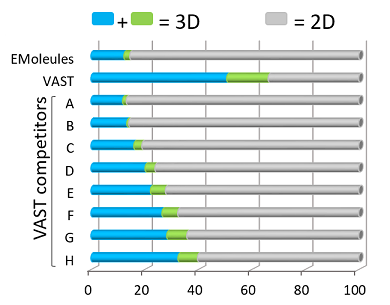
The 3D characteristic of VAST molecules is exemplified by an analysis performed by AstraZeneca (refer bar graphon the right). AstraZeneca's analysis accessed shape-diversity using principle moment of inertia (PMI) descriptors for a variety of chemical libraries including VAST screening library and VAST competitors offering 3D diverse libraries. Combining the blue and the green bars equates to compounds that are 3D in shape. It can be seen that more than 60% of VAST screening library is 3D in nature compared with the best VAST competitor of less than 40%.
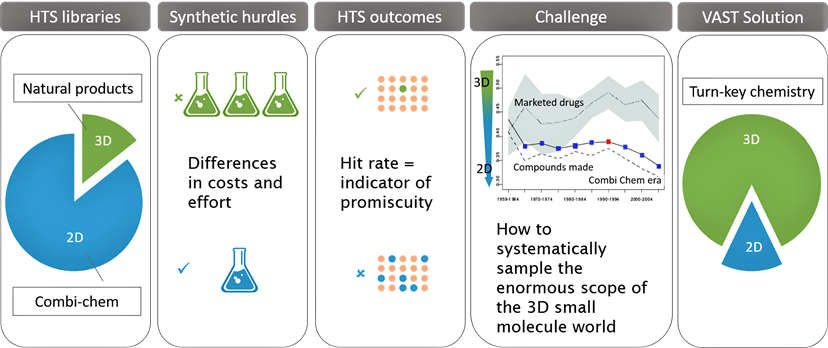
VAST technology addresses some of the hurdles faced by current HTS libraries.
The diagram to the left illustrates:
- HTS libraries have a small fraction of 3D molecules which are generally represented by natural products which are synthetically challenging. 2D molecules have less synthetic challenges compared with natural products, hence the over representation in HTS libraries
- The HTS hit rate for the 3D compounds is lower which translates to reduced promiscuity
- Marketed drugs are more 3D in shape compared with compounds found in HTS libraries
- Vast Bioscience has the solution with VAST technology - enabling the production of drug-like molecules using versatile turn-key chemistry
Vast Bioscience has a library containing more than 15,000 VAST molecules which it provides to partners for HTS.
VAST’s versatile chemistry provides unprecedented ability for optimisation. Unlike natural products, VAST technologyis versatile with established chemistry to access all five functional centres around the pyranose ring, allowing for practical optimisation of each fragment towards active leads with desired drug-like characteristics.
Vast Bioscience is now exploiting these opportunities through our business strategy in partnership with big pharma and through screening against in-house targets (pipeline).
PIPELINE
Vast Bioscience is independently pursuing a number of early stage projects aimed at identifying new clinical candidates for some of the pharmaceutical industry's most intractable targets resulting in unmet medical needs. Vast Bioscience intends to license these projects to pharmaceutical industry partners early in their clinical development.
Pain
Vast Bioscience is pursuing the development of a VAST based non-opioid pain therapeutic targeting both Nav1.7 and Nav1.8 . Pain is a major unmet medical need often poorly treated with opioids. The over use of opioids (or abuse) is now itself a major health threat.
Both Nav1.7 and Nav1.8 are voltage gated ion channels that have been genetically validated as pain targets. Vast Bioscience's pain project is in preclinical phase with oral efficacy in several in vivo pain models, including the post-surgical model (Brennan).
Antibacterial
New modalities to treat bacterial infections are needed with the increasing level of antibiotic resistance. The inhibition of the bacterial cell wall is a clinically validated pathway (as shown by beta-lactam class of antibiotics).
Glycosyltransferase (GTs) play a critical role in bacterial cell wall biosynthesis. Currently there are no drugs targeting GTs. Vast Bioscience is pursuing the development of GT inhibitors as a new-mode-of-action to treat drugresistant bacterial infections.
© Copyright www.vastbioscience.com
